03 Jul Olive Pests and Diseases
In the June edition of OliveCare® NEWS we profiled the Field Guide to Olive Pests, Diseases and Disorders in Australia (RIRDC 07-153), by Robert Spooner-Hart, Len Tesoriero, Barbara Hall.
https://www.agrifutures.com.au/product/field-guide-to-olive-pests-diseases-and-disorders-in-australia/
This publication is an old favourite for olive growers, summarising information on most of the possible pests, diseases and disorders occurring in the Australian olive industry. The field guide is designed as a quick reference and guide to take into the grove and use to identify pests and diseases and the damage they cause.
Note: This publication is currently being updated as part of olive levy funded project OL17001.
In this month’s OliveCare® News we explore major and minor olive diseases that occur in Australia, as well as some disorders of olives that can sometimes be confused with pest or disease damage:
Pest & Disease Symptoms – What to Look for:
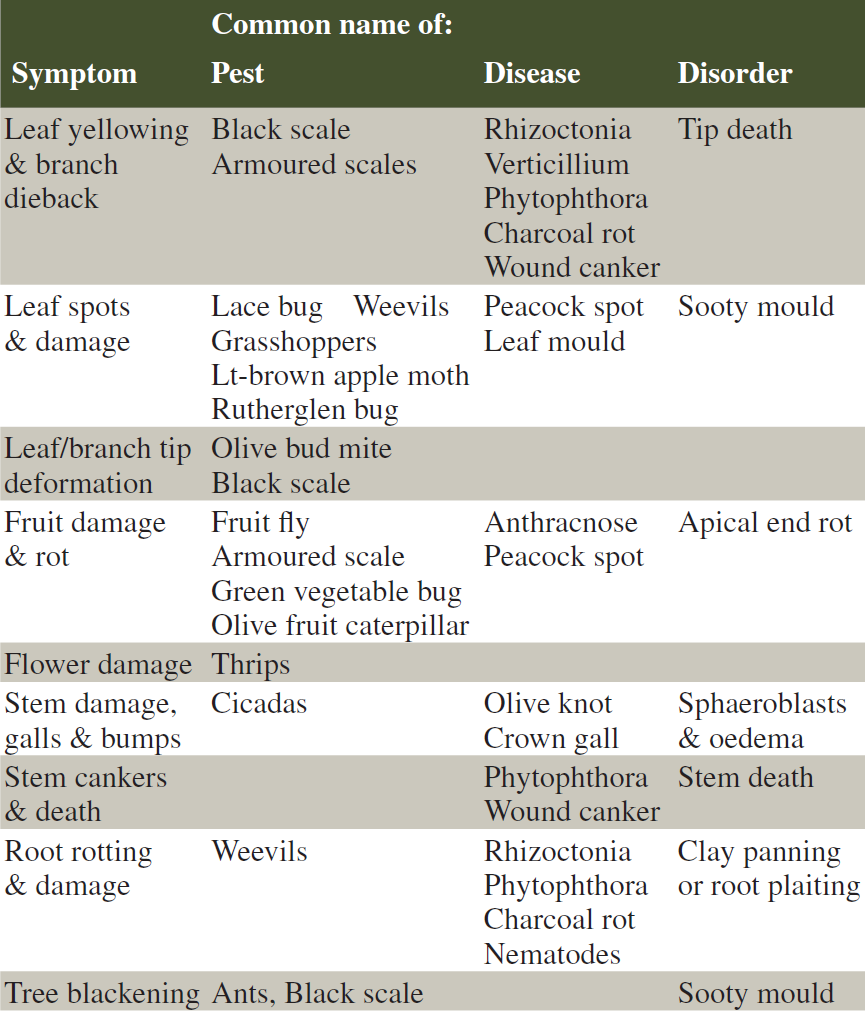
Use the table above to determine the possible causes of symptoms on your tree; the pest, disease or disorder can then be found alphabetically in the specific section of the publication based on common names.
Major Diseases of Olives
The following 3 fungal diseases are frequently observed in the olive tree canopy causing significant economic loss around the world, including in Australia:
Anthracnose:
Causal agents: Colletotrichum sp. (Colletotrichum acutatum and Colletotrichum gloeosporioides)
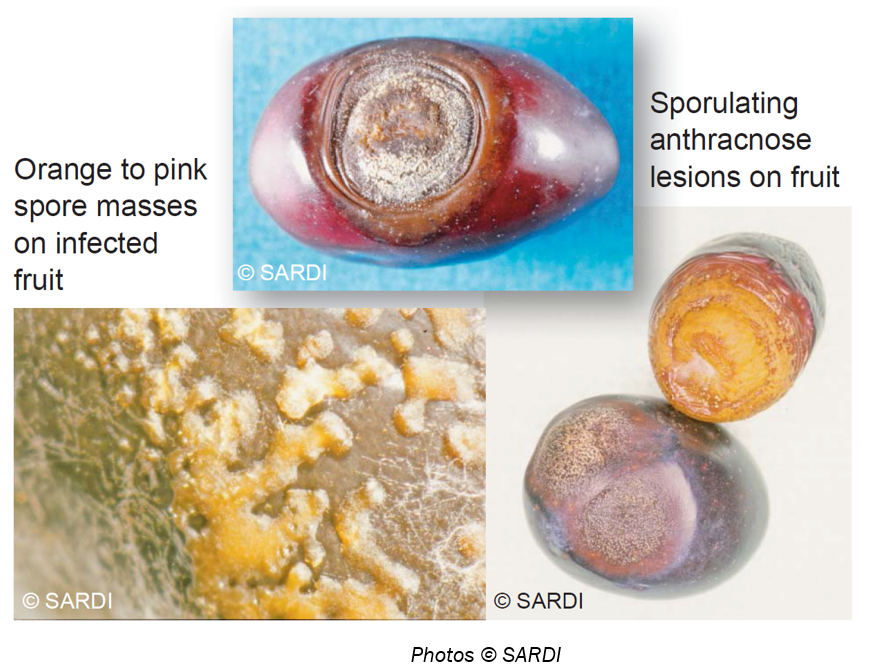

Background: Olive anthracnose is the most important fungal disease of olive fruits worldwide. It occurs in humid olive-growing areas of many production countries and causes heavy yield losses and lowering of oil quality. In Australia Anthracnose is a moderate to major problem depending on local climatic factors.
Symptoms: Causes soft circular rots on the fruit and at high humidity produces an orange coloured slimy mass of spores on the fruit surface; commonly observed close to harvest when the fruit softens.
Transmission: Favoured by wet conditions with high humidity, survives on infected mummified fruit. Spores are spread by rain splash and wind, can infect ripe fruit from new spores within 4 days.
Latent infections: To check if you have a latent Anthracnose infection in your grove, collect fruit samples in a plastic bag, add a little water and place in a warm location in your home or shed for a few days to incubate the spores and look for development of slimy orange spore masses on the olive skin.
Impact: Anthracnose infected flowers can lead to reduced fruit quality and premature fruit drop that reduces yield. When pressed the diseased fruit produce a turbid and acidic (High FFA) reddish oil of poor quality (defects: Musty / Humid / Grubby); the oil can be downgraded to a quality less than EVOO standard. Anthracnose is also problematic for table olives because of impact on taste and appearance.
Control: It is recommended that protective copper sprays be applied ahead of anticipated wet / high humidity weather conditions. Also after harvest remove all mummified fruit from the tree and cover fallen fruit and leaf trash with compost / wood chips / manure to prevent water splash reinfecting the new growth.
Grey Leaf Mould:
Alternative names: Cercospora leaf mould, olive leaf spot, cercosporiosis.
Causal agent: Fungus: Pseudocercospora (= Cercospora = Mycocentrospora) cladosporioides
Ref: http://olivediseases.com/media/Olive-diseases-in-Australia-Power-point-presentaion-copy2.pdf
Transmission Not all infected leaves fall from the tree, and the fungus overwinters in old infected leaves. Usually infects tree in the autumn affecting the young spring growth.
Favoured by High humidity and rain, temperatures of 12–28 °C.
This disease seems to spread at lesser humidity than for peacock spot, so it occurs more frequently in warmer areas, especially after humid and rainy spring weather.
Control: This fungus infects the lower surface of the leaves, which is also more protected from spray treatments, meaning treatments need to be more radical than for peacock spot – prune and thin the tree canopy and use preventative and curative sprays.
The internationally favoured control option for Grey Leaf Mould is Mancozeb a Group M3 fungicide, however this product is not yet registered for use on olives in Australia; this means Australian olive growers (other than those in Victoria) are currently restricted to the use of copper formulations for the control of Grey Mould (see following label and permit details).
http://permits.apvma.gov.au/PER11360.PDF
http://www.herbiguide.com.au/Labels/CUOXY520_51820-0708.PDF
Also download the following helpful guide to using copper sprays to control olive diseases by Australian plant pathologist Dr Vera Sergeeva: http://olivediseases.com/media/vera_copper_sprays.pdf
Symptoms The grey mould can be seen mostly on mature leaves, with mouldy blotches developing on the underside of the leaves. The tops of the leaves turn yellow then brown, leaves then fall. Often occurs together with peacock spot, causing significant defoliation and damage to new growth and reduced crop production.



Cercosporiosis on olive fruits:
P.cladosporioides has been considered primarily a leaf pathogen, with fruit rarely infected, if so, fruits show round, reddish-brown spots. Outbreaks can be sporadic, and the disease may take several years before it becomes serious.

Peacock Spot:
Alternative names: scab, olive leaf spot, bird’s eye spot)
Causal Agent: A fungus Spilocaea oleagina (= Fusicladium oleagineum).
Transmission: Fungus overwinters in old infected leaves. Spores germinate in free water and are blown or splashed onto the leaves. Movement between trees is limited.
Favourable conditions: Key infection periods are Spring and Autumn following periods of leaf wetness (optimum 2 days wetness at between 15-20 degrees C). Incubation periods vary from 10 days to several months after the initial infection with development delayed by a cold winter or hot summer before the spots form on the leaves in late Summer to early Autumn. The pathogen can also survive for over 200 days as conidia on infected leaves that remain symptomless on the tree, and are subsequently carried over short distances primarily by rain in the direction of the prevailing winds to reinfect the canopy when environmental conditions are favourable. Because of this long latency period the disease, initial infection is often missed waiting for the characteristic spots to appear.
These microclimatic conditions of high ambient humidity and mild temperatures occur mostly in spring and autumn, particularly in closely planted groves with poor canopy aeration, and in groves situated close to water courses, in valleys and other moist locations. Rain is not the issue as this can actually wash spores form the leaf, rather it is when the leaves stay wet.
For the spore to develop into an infection, moisture must remain on the leaves for several hours with mild temperatures. Usually the disease occurs sporadically, particularly in wet weather in spring. Disease is inactive during summer.
Symptoms of Peacock Spot: Symptoms are characterised by ‘peacock’s eye’ shaped spots of 2-10mm in diameter mainly on the upper surface of the leaf, and occasionally on the stems and fruits. Spots first appear as small pale blotches, later becoming muddy green to black, often with a yellow halo. Spots on underside of leaves are grey. Young leaves may remain symptomless.
The primary impact of the disease is on reducing photosynthetic efficiency of the infected leaves. Severe infection may cause defoliation and associated loss of accumulated minerals and photo-assimilates and death of new wood, which reduces production (less flower bud initiation, fruit, smaller fruit and lower oil accumulation) in the following year.



Control: Is through the use of tolerant / resistant varieties, lower tree planting density, open up tree canopy to improve aeration; avoid overuse of nitrogenous fertilizers that increase disease susceptibility of the leaves; and the timely application of copper preventative sprays which prevent the spores from germinating (usually applied later summer and late winter) corresponding to the main infection periods, in severely affected groves an additional copper treatment in late spring may also be necessary. Modern translaminar (curative) sprays are also now available but not yet registered for use on olives.
Note: High temperatures and low humidity of summer prevents the spores from germinating. In winter low temperature conditions also prevent the spores from germinating.
A note on apical end rot:
Alternative names Apical end desiccation, soft nose
This includes physiological disorders of olive fruits that can be confused with anthracnose.
Symptoms The apical (blossom) end of the fruit shrivels, mostly close to maturity. The internal flesh and pip may be blackened, either at the apical end only or throughout the whole fruit. Sometimes secondary fungal rots infect the shrivelled end.
Causes: It may result from sudden changes in temperature and humidity which produce partial dehydration of the fruit at the apical end. It has also been associated with calcium and boron deficiencies, and with changes in watering regimes.

A note on sooty mould:
Sooty mould is NOT a leaf fungal infection
Causal Agent: Fungi: Capnodium sp. (most common); also Fumago, Scorias & Aureobasidium spp.
The fungal spores are wind blown and attach to the honeydew excretions from sap-sucking insects, particularly black scale but also aphids, mealybugs and psyllids.
Symptoms: A black soot-like growth which can cover all surfaces of the plant. Severe infections can indirectly cause plant stunting and un-thriftiness, as the soot coverage prevents sunlight penetration and thus compromising photosynthesis by the plant.
Control: To manage sooty mould, the insects producing the honeydew must be controlled.

Other Fungal Diseases of Olives
The following fungal diseases are soil born, and less frequently observed in the olive grove, but can cause serious loss of productivity and economic loss:
Verticillium Wilt:
Causal agent: Verticillium dahlia (non-defoliating strain)
Symptoms One or more branches wilt, usually early in the growing season. Dead leaves remain on the tree. Roots are repeatedly infected over several seasons and trees gradually die. Internal tissue of lower stems may darken as the fungus disrupts the ring of sap-carrying tissue under the surface. Olive cultivars vary greatly in susceptibility, and symptoms may not be seen for 4 to 8 years after planting.
Transmission: Soil-borne. The fungus survives in soil for many years and can infect the roots of a wide range of plants. It can spread in irrigation water and infected soil on farm machinery and tools.
Favoured by Cool and moist soils when daytime temperatures range between 20 and 25 °C. Suppressed by higher temperatures. Common in land where alternate hosts (e.g. cotton, lucerne, brassicas) have been grown.
The Verticillium Wilt fungus affects the vascular system starting in the soil from spores close to a root.
Note: a whole branch is usually affected (indicating the infection is moving up the tree xylem from one or more of the infected roots. This wood should be removed and burnt.
The fungus infests the tree from the roots to the leaves, and dead leaves fall to the ground containing spores to continue the cycle of infection.
Damp clay soils with high temperature are especially favourable for infection, such as in irrigated groves. Soils high in organic matter are resistant to Verticillium due to the presence of antagonistic microorganisms. Use of compost is therefore recommended.
Control: There is no way to eradicate the infection from a tree, although the tree can be nursed along with reduced irrigation and reduced nitrate fertilizers, pruning and balanced nutrition. Management is based on avoidance of the fungus and reducing inoculum level in the soil:
- Use disease free planting material
- Plant resistant varieties
- Avoid planting into ground previously planted to alternative hosts of the fungus eg cotton, stone fruit, Brassica’s, potatoes and tomatoes.
- Avoid inter-row cropping with susceptible plants, eg clover
- Avoid soil movement from infected areas to non-infected areas
- Reduce inoculum levels before replanting by keeping the soil weed free and growing resistant plants eg grasses for several years. Fumigation of the soil will also reduce inoculum.
Ref: https://apsjournals.apsnet.org/doi/pdfplus/10.1094/PDIS-06-11-0496


©NSWDPI: Darkened ring of water-conducting tissue inside stem
Biosecurity Note: Verticillium dahliae (exotic defoliating strains) – a more serious form of this fungus causes tree defoliation and death. This is already present in Australia as a disease of cotton, but not yet reported on olives.
Ref: https://www.planthealthaustralia.com.au/pests/verticillium-wilt-defoliating-strain/
A Note on Tip Death
Symptoms Ends of branches die for no apparent reason. Tip death appears to have no effect on the general health of the tree or its productivity. Branches can be removed if this is considered necessary for cosmetic purposes. Inspection of the stem below the dead tips is needed to determine whether the death has a specific cause which should be further investigated (see Wound Cankers, p48). Root rot and trunk cankers can also cause tip death, and so should be investigated.

Phytophthora Root Rot:
Causal agent: Phytophthora spp. (7 species have been identified as affecting olives)
Symptoms: Root and crown cankers that may extend up the trunk. Leaves of infected trees become yellow and drop, stems wilt and die back from the tips. The tree usually shoots from dormant buds lower down on the tree, either from the main trunk or lateral branches. Vascular browning primarily of the xylem tissue is usually present in affected parts of the tree. Cracking and distortion of the trunk can also occur. Trees may die suddenly, or slowly decline over several years. Sudden death is common when stress is placed on the tree, such as during flowering, fruit development or hot weather.
Transmission: Phytophthora is a soil borne organism, spread by movement of soil, water and infected plants.
Favoured by: Phytophthora root rot is consistently associated with excessively wet soils, clay-panning or poor soil drainage. Care must be taken when using feral plants as rootstocks, as many grow in areas where Phytophthora is present in the soil.
Control: Avoid water logging and excessive irrigation. Implement grove drainage systems – an important consideration in grove design. Avoid soil movement from infected areas (including on vehicles and machinery)

Ref: http://www.australisplants.com.au/olives/rootrot.htm
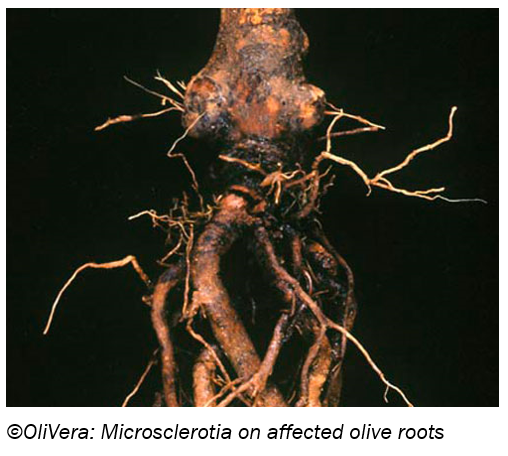
Charcoal rot:
Causal agent: Macrophomina phaseolina (also called Rhizoctonia bataticola)
Ref: http://olivediseases.com/media/vera_macrophomina_olives.pdf
Tree decline caused by Macrophomina phaseolina (Tassi) Goid., a disease of olives detected in the drier areas of Southern Australia (Sergeeva et al., 2005).
Symptoms Plants die back from the shoots, and leaves drop. Affected roots appear grey and are dotted with tiny (pinhead-sized) black speckles (microsclerotia) on their surface these are survival structures of the fungus. Severely affected roots blacken and rot away. Leaf symptoms develop when plants are heat stressed.
Transmission: Soil borne, this fungus survives in soil for many years and can infect roots and stems of a wide range of plants. It spreads in irrigation water and infected soil on farm machinery.
Favoured by: Unlike Phytophthora this fungus favours drier soil conditions and is associated with root rots occurring where plants have been water-stressed during summer.
Control: Avoid water stressing olive trees.
Bacterial disease of olives
Olive Knot
Causal agent: Pseudomonas savastanoi pv. Savastanoi
Ref: http://ipm.ucanr.edu/PMG/PESTNOTES/pn74156.html
Olive Knot is a bacterial disease of olives and can seriously affect production. ‘Olive Knot’ is spread through contact with tree wounds caused by hail and wind; damage by harvesting and pruning equipment. Olive Knot has not yet been detected in all states, so if you see symptoms, notify your state agriculture dept.
Symptoms: Rough galls or swellings of variable size occur on twigs, branches, trunks, roots, fruit or leaves. Galls can appear either singly or close together. They are most common on twigs and young branches, but will also form around wounds on the main trunk. Starting as small swellings 3 to 5 mm across, they grow rapidly into smooth, spherical green knots, increasing in size as they mature and becoming darker and more furrowed.
Transmission: The bacteria are present in the gall releasing a hormone that forms the gall tissue. When the tree is wet, the bacteria can migrate and start up new knots. The bacteria can’t infect healthy bark, rather they enter through recent wounds, including leaf scars, damage by hail and frost, from branches rubbing in the wind, pruning wounds or wounds caused during harvesting.
The galls or olive knots occur mainly on the thinnest branches, impeding sap flow so these branches lose vigour and productivity.
Favoured by: Trees at most risk are those with wounds during periods of rain. Some cultivars are more susceptible, e.g. ‘Barnea’, ‘Frantoio’.

©ResearchGate: Galling on olive branches around leaf abscission points
Control measures: Avoid pruning and harvesting in wet weather; disinfect pruning and harvesting tools and machinery; remove infected plant material from the grove; use copper sprays on recently pruned and harvested groves to impede new infections, (these will not control existing infections); apply copper sprays immediately after hail storms; plant resistant varieties, Arbequina variety is particularly susceptible to olive knot.
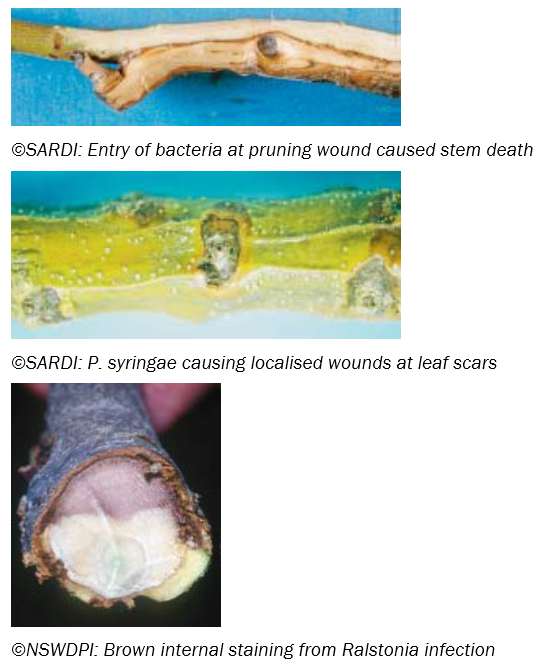
Other Wound Cankers
Causal agents: Opportunistic wound invaders – Fungi: e.g. Botryosphaeria sp., Pycnoporus coccineus (white wood rot); Bacteria: e.g. Pseudomonas syringae, Ralstonia solanacearum, Xanthomonas campestris
Symptoms: Vary from slow decline of trees and tree death to localised cankers around wound sites with occasional branch death above infection. Can also cause brown staining of the vascular (sap-carrying) system.
Transmission: All are wound pathogens. Can be borne by wind, water and soil. Most are common organisms that opportunistically infect through wounds.
Favoured by: Wounds, wetness and high humidity causing moisture films around wound sites.
Control: Avoid causing wounds to olive trees
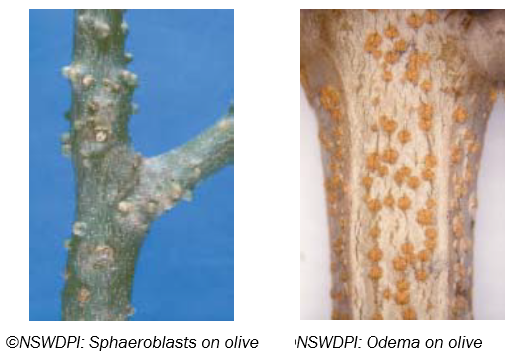
A Note on Sphaeroblasts and Oedema
These are benign structures likely resulting from tree physiological reactions, which may be confused with disease cankers:
Sphaeroblasts are knob-like growths up to 10 mm wide which protrude from stems. When they are cut open, a spherical lump of wood can be removed from the surrounding tissue. Their cause is unknown, and they commonly occur on the cultivar ‘Barnea’.
Symptoms of oedema are small, brown, corky growths up to 5 mm wide that form on the surface of stems or roots from enlarged lenticels (breathing holes in bark). They occur when high soil moisture causes excessive water uptake, which engorges the cells near the lenticels. These cells can rupture from the high water pressure, and the plant forms callus tissue in an effort to heal. When roots experience periods of high soil moisture, some tissue may also asphyxiate (because of reduced oxygen levels). Consequently, these roots are predisposed to infection by a range of minor pathogens or opportunistic invaders such as Fusarium, Pythium and various bacteria.
What is your experience with fungal and bacterial diseases in your grove?
Olive growers are invited to share their observations and personal experiences with fungal and bacterial diseases of olives. Are any of the above diseases and disorders a problem in your grove?
If you have experienced any of these diseases and disorders, do they vary in impact between varieties, tree age, grove practices?
Do you use IPDM strategies such as promoting soil and tree health?
If you use chemical control options – what works and what doesn’t work?
Please send your observations / comments to olivecare@australianolives.com.au
Pest & Disease Control Options
Remember that ‘happy trees are healthy trees’ – pests and disease outbreaks may indicate your olive grove is under stress – humid weather, water stress, soil nutrient deficiencies, tree canopy out of control, natural beneficial organisms present in low numbers, etc.
Spray carefully, if you must spray
- Understand pest and disease life-cycles:
- Spray if really necessary? healthy trees, few problems?
- One Spray enough? Can you limit to spot-sprays?
- Catch early – sprays will be more effective
- Understand the Spray and its effect:
- Is it legal in your state?
- Does it kill beneficials – even if it is organic?
- When do you spray again – critical life cycle times?
Be sure to check out the approved chemicals for use on olives:
Ref: https://australianolives.com.au/chemical-permits/
Other useful references:
Olive Growing (El Cultivo Del Olivo) Ch 15: Diseases by Antonio Trapero and Miguel Angel Blanco: https://australianolives.com.au/olive-growing-manual/
EIP-AGRI Focus Group ‘Pests and Diseases of the Olive Trees’ 28 May 2020 Sergiu Didicescu: https://ec.europa.eu/eip/agriculture/en/publications/eip-agri-focus-group-pests-and-diseases-olive-tree-0
Olive Diseases and Disorders, Ed Schena L. et al Transworld Research Network 2011
Evaluation of Key Chemicals for Pest Management – Robert Spooner-Hart, Vera Sergeeva UWS: https://australianolives.com.au/wp-content/uploads/2019/05/SPOONERHART-Robert.pdf
